What are the Different Generations of Solar Cells?
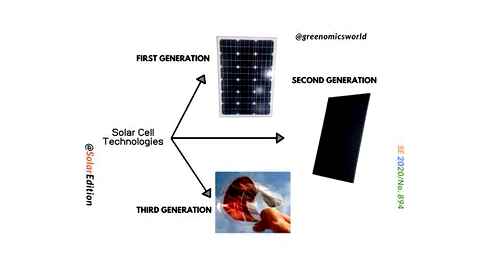
There are three basic generations of solar cells, though one of them doesn’t quite exist yet, and research is ongoing. They are designated as first, second, and third, and differ according to their cost and efficiency.
The first generation are high-cost, high-efficiency. These solar cells are manufactured in a fashion similar to computers, involving extremely pure silicon, use a single junction for extracting energy from photons, and are very efficient, approaching their theoretical efficiency maximum of 33%. In 2007, first generation products accounted for 89.6% of commercial production, though the market share has declined since. The manufacturing processes that are used to produce them are inherently expensive, meaning that these cells may take years to pay for their purchasing costs. It is not thought that first generation cells will be able to provide energy more cost effective than fossil fuel sources.
The second generation, which has been under intense development during the 1990s and early 2000s, are low-cost, low-efficiency cells. These are most frequently thin film solar cells, designs that use minimal materials and cheap manufacturing processes. The most popular materials used for this type are copper indium gallium selenide, cadmium telluride (CdTe), amorphous silicon, and micromorphous silicon.
A standard example of second generation cells would be those made by Nanosolar, which uses a special machine to print the cells at an extremely fast rate. Though these cells have only 10-15% conversion efficiency, the decreased cost more than makes up for this deficit. Second generation cells have the potential to be more cost effective than fossil fuel.
Third generation solar cells are just a research target and do not really exist yet. The goal of solar energy research is to produce low-cost, high efficiency cells. This is likely to be thin-film cells that use novel approaches to obtain efficiencies in the range of 30-60%. Some analysts predict that third generation cells could start to be commercialized sometime around 2020, but this is just a guess. Technologies associated with third generation products include multijunction photovoltaic cells, tandem cells, nanostructured cells to better pick up incident light, and using excess thermal generation to enhance voltages or carrier collection.
Michael is a longtime TheSolarPanelGuide contributor who specializes in topics relating to paleontology, physics, biology, astronomy, chemistry, and futurism. In addition to being an avid blogger, Michael is particularly passionate about stem cell research, regenerative medicine, and life extension therapies. He has also worked for the Methuselah Foundation, the Singularity Institute for Artificial Intelligence, and the Lifeboat Foundation.
Michael is a longtime TheSolarPanelGuide contributor who specializes in topics relating to paleontology, physics, biology, astronomy, chemistry, and futurism. In addition to being an avid blogger, Michael is particularly passionate about stem cell research, regenerative medicine, and life extension therapies. He has also worked for the Methuselah Foundation, the Singularity Institute for Artificial Intelligence, and the Lifeboat Foundation.
Recommended
AS FEATURED ON:
AS FEATURED ON:
Discussion Комментарии и мнения владельцев
@Georgesplane- I study renewable energy and have read up on these systems so I can answer your questions. These new solar cells are not going to be s cheap as the solar cells the CPV manufacturers were using before, but they are more than double their efficiency.
CPV systems can also concentrate solar radiation up to 1000 times, which is double what they were capable of two or three years ago. This allows the systems to use fewer cells (about half of current and about a quarter of what would have been necessary a few years ago) for a collector while not sacrificing output.
As for residential use, these systems will be strictly grid tied commercial generation plants. They will be used for plants rated at about 50MW, give or take. They will not necessarily make the cost of generated electricity more as long as the increase in cost does not surpass the percentage increase in efficiency. This technology improvement will help the industry mature and eventually drive down costs. Georgesplane yesterday
@Amphibious54- What are the costs of this third generation solar cell technology compared to the costs of the cells the CPV manufacturers were using before? Has the company manufacturing these cells delivered a cheaper product with increased efficiency as many in the industry have touted this technology before? Will these CPV systems be available for residential use, and if so, how much will they cost per installed kW? Does the technology compete with electricity from centralized generation plants? Amphibious54 May 26, 2011
I would just like to add that a company announced earlier this year that they will start full-scale, commercial production of third generation cells by the beginning of next year. The company has produced a cell that is rated at a record holding 41.4% efficiency.
The cell is basically a number of layers of semi conductive material that each absorb a certain wavelength of light. The wavelengths that each the first layer does not absorb is passed through to the next layer and so on. Each layer is then organized in series to convert the solar radiation into electricity.
The cells are meant to work with Concentrating Photovoltaic (CPV) systems. This means they will not be sold directly to the public, but to other companies that produce CPV systems. The company plans to produce 250,000 cells per year if it receives the grant from the Department of Energy for production expansion.
Solar Cells
Solar cells, or photovoltaic (PV) cells to give them their correct name, are used to convert light energy into electrical energy. The appeal of a solar cell is the promise of a clean source of energy that does not generate any harmful by-products, like greenhouse gasses.
Whilst solar cells require sunlight to provide efficient levels of power generation GCell can use sunlight or artificial ambient light to create electrical energy. However, all solar cells use the same basic construction of using a semiconductor to conduct electricity under certain conditions.
When a photon (a particle of light) hits the photo-sensitive or light-active component of the solar cell it excites the electrons allowing them to flow freely. The electric fields within the solar cell push the excited electrons to move in a certain direction. This movement of electrons is current (ampere).
Placing metal contacts on the top and bottom of the solar cell, can direct the current for use externally. Together with the solar cell’s voltage (which is determined by its built-in electric field), it defines the power (wattage) that the solar cell can produce.
Different types of solar cell
In the below sections we look at the different types and generations of solar cells.
1st generation solar: crystalline solar cells
The 1st generation of solar cells, typically wafer-based, date back to 1958 where they were used in the first satellites launched into orbit. Today now that they are more affordable they are widely used on rooftops and free-standing solar-farm systems around the world.
Typically the 1st generation solar panels provide the most power per square metre of surface area under standard test conditions (STC); Temperature 25°C, Irradiance 1000 W/m2 (1sun), Air mass 1.5 (AM1.5) spectrum. However, low-light conditions and rising temperatures impact performance.
Being silicon-based they are rigid solar panels that are brittle and fragile, not good for transporting or handling. They have extensive lifetime of 10-20years and can handle exposure to the elements very well. They are also one of the most energy intensive photovoltaic technology’s to manufacture because very pure silicon is required.
Good outdoor performance Good outdoor lifetime – Poor indoor / low-light performance – Energy intensive manufacture – Worst physical characteristics
2nd generation solar: thin-film solar cells
The 2nd generation of solar panels include amorphous silicon (a-Si), Copper Indium Gallium Selenide (CIGS) and Cadmium Telluride (CdTe). Amorphous silicon cells date back to 1976 and were used on the world’s first solar power calculator the EL-8026 from Sharp. Today 2nd generation solar cells are used in a wide range of application from rooftops to textiles.

Thin-film, so called because they use semi-conductor materials only a few micrometers thick, means they can be manufactured on substrates other than glass. This makes 2nd generation solar cells light-weight, easier to integrate and cheaper to manufacture.
However, some 2nd generation solar cells use toxic materials and vital raw materials that are in great demand in other applications, for example flat screen displays, which could lead to long-term price increases.
Cheaper than 1st generation solar cells Light-weight and easy to integrate – Average outdoor performance – Poor indoor / low-light performance – Uses toxic or rare earth materials
3rd generation solar: organic solar cells
The 3rd generation of solar cell includes dye-sensitized solar cells (DSSC) and organic photovoltaics (OPV) which use organic dyes and organic polymers respectively, to create a photoexcited electron from which energy can be harvested.
The creation of DSSC dates back to 1991 and has been commercially available since 2006 through G24. With its indoor attuned chemical formulation GCell, by G24 Power, focuses on indoor applications where robustness, flexibility and easy integration are ideally suited to the growing portable wireless consumer electronics market.
The future prospect of roll-to-roll manufacturing, cheaper materials, more stable molecular dyes and quantum dots, transparent and semi-transparent coloured solar cells, and greater power density creates opportunities for the use of GCell in large scale electrical solar power generation such as glass panes or cladding for buildings.
Superior indoor / low-light performance Light-weight, flexible, robust and easy to integrate  Prospects of lower-cost, long-life and greater power density. – Short-term life expectancy ( 5-years)– Poor direct sun performance (100,000lux)
Bimetallic Implanted Plasmonic Photoanodes for TiO2 Sensitized Third Generation Solar Cells
An auspicious way to enhance the power conversion efficiency (PCE) of third generation sensitized solar cells is to improve the light harvesting ability of TiO2 sensitizer and inhibition of back recombination reactions. In the present work, we have simultaneously comprehended both the factors using stable bimetallic Au and Ag metal nanoparticles (Mnps) embedded in TiO2 with ion implantation technique at lower fluence range; and explored them in third generation dye sensitized solar cells (DSSCs). The best performing Au-Ag implanted DSSC (Fluence- 6 × 10 15 ions cm −2 ) revealed 87.97% enhancement in its PCE relative to unimplanted DSSC; due to plasmon induced optical and electrical effects of Mnps. Here, optimized bimetallic Au-Ag Mnps embedded in TiO2 improves light harvesting of N719 dye; due to the well matched localized surface plasmon resonance (LSPR) absorption Band of Au and Ag with low and high energy absorption bands of N719 dye molecules, respectively. Furthermore, Au and Ag acts as charge separation centers in TiO2 that inhibit the recombination reactions occurring at photoanode/electrolyte interface via prolonging photo-generated electron lifetime; resulting in efficient inter-facial charge transportation in DSSCs.
Introduction
In last decade, third generation sensitized solar cells have achieved tremendous consideration out of authoritative traditional silicon based photovoltaic technology in tandem cell configuration; owing to their low manufacturing cost, non-toxic nature and undeniable higher theoretical limit of power conversion efficiencies (PCE) 1,2,3,4,5. Sensitizer plays a key role in third generation sensitized solar cells; thus should exhibit phenomenal properties like high chemical and thermal stability, high photo-catalytic activity, low-cost, high redox ability, strong absorption coefficient, biocompatibility, high specific surface area, non-toxicity, and recyclability 6. TiO2, an extrinsic n-type semiconductor, fulfills all the requirements and hence widely studied as sensitizer in third generation photovoltaic devices constituting quantum dot sensitized solar cells, dye sensitized solar cells (DSSCs), and pervoskite sensitized solar cells 7. However, TiO2 appears to undergo some limitations such as random charge transportation occurring due to trapping and detrapping of photo-generated electrons in its trap levels resulting in recombination losses and light absorption confined only in UV region of solar spectrum; hence requires some modifications which can be done via incorporating different shape, size and dimensions of TiO2, metal oxides, metal nanoparticles (Mnps), hybridized carbon materials, and coupling with semi-conducting materials 8,9,10. Among them, inclusion of Mnps such as gold (Au) and silver (Ag) induce plasmonic optical and electrical effects in TiO2, due to their unique localized surface plasmon resonance (LSPR) property; which effectively strengthens their light absorption and charge transportation ability and ultimately improves the PCE of sensitized third generation solar cells 11,12,13.
Recently, we observed that the single crystalline spherical shaped Ag nps incorporated in TiO2 via chemical reduction method, effectively improves the absorption cross-section of dye sensitizer 14. over, the inclusion of different single crystalline anisotropic shapes of Ag nps in TiO2 results in improved light harvesting ability of TiO2; as they exhibit multiple LSPR bands in the broadened region of solar spectrum ranging from 380–900 nm 15. Although, bare Mnps significantly improves the light harvesting ability of TiO2 via enhancing the absorption cross-section of dye sensitizer; they become unstable coming in contact with liquid based electrolytes and gets corroded.
Xu et al. demonstrated the utilization of Au-Ag alloy popcorn shaped core-shell nanoparticles in TiO2 to broaden the light absorption range via using efficient excitation of LSPR modes on the popcorn nanoparticles 16. However, core-shell structures improved the stability of Mnps in liquid electrolytes, but suffered from their unpronounced LSPR effect on light harvesting ability of TiO2.
Furthermore, ion implantation have been observed to be practically quite effective technique to embed Mnps in TiO2; which helped in resolving both the instability and unpronounced LSPR issues of bare and core-shell structures, respectively 17,18. Recently, we have explored the ion implantation method to implant Au Mnps in TiO2 and employ them as photoanodes in third generation DSSCs. The optimized Au implanting fluence provides balancing effect of LSPR of Au and effective adsorption area of TiO2 for N719 dye in DSSCs; and showed an enhancement of 44.7% in its PCE relative to unimplanted TiO2 based DSSCs 19. Similarly, Ag Mnps implantation in TiO2 showed 65.3% enhancement in PCE of DSSC 20. However, their increment is not a match for its economical utilization; since efficient light harvesting in Au as well as Ag implanted DSSCs is only around single LSPR absorption wavelength; resulting in unsatisfactory PCEs.
In order to further enhance the light harvesting ability of TiO2 within the whole visible region of solar spectrum, Kim et al. demonstrated the efficient energy matching between the absorption bands of N719 dye and Au and Ag Mnps; resulting in relatively enhanced PCE of double layered plasmonic DSSCs by 19.12% 21. Wang et al. introduced the plasmonic cooperation of Au and Ag Mnps in TiO2 photoanodes of DSSCs using chemical reduction method and achieved 20.8% PCE enhancement; by exploiting the strong plasmonic cooperation effects due to the complementary light absorption of both the Au and Ag Mnps via their respective LSPR absorptions at ~550 and 400 nm 22. Yun et al. demonstrated the incorporation of core shell Au@Ag Mnps in TiO2 hollow spheres leading to synergistic effects of improved light harvesting and widening of absorption Band, appearing due to effective light scattering effects of core-shells; which raised the PCE by 25% 23. Dong et al. proclaimed 40% increment in PCE of DSSCs comprising Ag-encapsulated Au nanorods prompting enhanced light harvesting and efficient one dimensional charge transportation 24. Al-Awazi et al. showed the optimal concentration of Au-Ag alloys (4:1) introduced into TiO2 via pulsed laser ablation, emanating PCE by 52.1%; arising from the broader optical absorption of dye molecules using plasmonic effects of Au and Ag Mnps generating larger number of photo-generated charge carriers 25. A seed mediated growth of Ag shells on Au Mnps incorporated in TiO2 photoanodes have been investigated by Salimi and his co-workers, and noted 125% enhancement in PCE of DSSCs due to their prominent light absorption as well as enhanced inter-facial charge transportation through Mnps minimizing the charge recombination processes 26. Till now, maximum 230% enhancement in PCE of DSSCs, having modified TiO2 photoanodes with Au-Ag nanocomposites, have been achieved by Lim et al. that originates from LSPR synergistic interactions between Au and Ag Mnps resulting in the improved light harvesting; and efficient charge separation and transportation processes 27.
Above reported literature mentioned the simultaneous inclusion of Au and Ag Mnps in photoanodes of DSSCs using chemical methods, and their PCEs enhancement is not at par with the theoretical maximum value; resulting from the uncontrolled growth and concentration of Mnps which leads to their non-linear conglomeration in TiO2; hence deteriorating their long term stability and sustainability. In this direction, we have investigated the use of ion implantation technique to embed Au and Ag Mnps inside TiO2 semiconductor of DSSCs; resulting in efficient enhancement in the PCE of highly stable plasmonic DSSCs via extending the region of light harvesting within UV-Vis-NIR, by utilizing LSPR absorptions of Au and Ag Mnps as well as improving the charge transfer processes by reducing recombination rate of photo-generated charge carriers, owing to their charge storage ability.
Results and Discussion
Concept visualization
Bimetallic Au and Ag implantation in TiO2 will significantly improve its light harvesting ability and inhibit back recombination reactions, resulting in better electron transportation; that positively influences the photovoltaic performance of DSSCs. Figure 1 represents the schematics of electron transportation processes occurring in DSSCs using bimetallic Au-Ag implanted TiO2 photoanodes. When light falls on the photoanode of DSSCs, enormous photo-generated electrons are produced from excited N719 dye molecules, especially from the ones present in the vicinity of Au and Ag Mnps. Rapid accumulation of photo-generated electrons on Au and Ag Mnps takes place due to their charge storage ability (Fig. 1(1)); which in turn induces an upward shift in the Fermi energy level (EF) of photoanode; resulting in efficient transportation of accumulated photo-generated electrons to the conduction Band (C.B.) of TiO2 (Fig. 1(2)) 28. over, the trap levels of TiO2 are filled by the electrons generated from the plasmonically excited Au and Ag Mnps, which reduces the random charge distribution of photo-generated electrons in TiO2; hence inhibits the recombination reactions occurring at photoanode/ electrolyte interface (Fig. 1(3)) 29. Further, the photo-generated electrons from the C.B. of TiO2 get transported to the external circuit via TiO2 compact layer deposited FTO and are collected at Pt CE 30. Thus, Au and Ag Mnps in TiO2 would efficiently enhance the charge transportation processes throughout DSSC via elongating electron lifetime as well as transport path length. The above mentioned concept exhibiting higher light harvesting ability as well as better charge transportation processes in bimetallic Au and Ag implanted photoanode based DSSCs has been fully supported by the underlying investigations.
Structural and Optical characterization of photoanodes
XRD diffraction patterns of unimplanted and Au-Ag implanted TiO2 on FTO substrates are depicted in Fig. 2. TiO2 exists in both the anatase (A) and rutile (R) phase with peaks appearing at 26.08°, 27.22°, 38.42°, 48.66°, 54.96°, 64.48°, and 66.18° Bragg’s angle relating to (110)-R, (110)-A, (004)-A, (200)-A, (211)-A, (116)-A, and (204)-A crystal planes, respectively (JCPDS Card No. 21–1272 and 04-0551). In case of Au-Ag implantation in TiO2, two new crystal planes (200) and (311) at 44.04° and 77.58° Bragg’s angle, respectively appeared along with slight increase in the intensity of (004), (211), and (116) plane of TiO2; which is due to the overlapping of Au, Ag and TiO2 crystal planes; as they exhibit very similar lattice constants that further confirms their metallic face centered cubic crystal structure (JCPDS Card No. 04-0784 for Au and 04-0783 for Ag). The peak intensities with overlapped Au, Ag and TiO2 crystal planes enhances initially with increasing Au-Ag implantation fluence, that results in the formation of large number of metal nanoparticles (Mnps); but then suddenly decreases which is due to the suppression of TiO2 planes as they get covered with Au and Ag Mnps as well as thin TiO2 melt. Thus, the crystallinity of Au-Ag implanted TiO2 increases particularly along (200) and (311) crystal planes; leading to better charge carrier mobility which is necessity for improved DSSCs.
The elemental composition as well as chemical state of unimplanted and Au-Ag implanted TiO2 at varying fluence are analyzed from the XPS survey spectra demonstrated in Fig. 3; which shows four peaks of Ti-2p, O-1s, Au-4f and Ag-3d elements. The high resolution Ti-2p spectra of TiO2, present in all the samples (Fig. 4); exhibited two peaks centered at binding energies 458.3 and 464.3 eV corresponding to Ti-2P3/2 and Ti-2P1/2, respectively; which indicates the presence of Ti 4 oxidation state 27. O-1s peak observed at binding energy 529.9 eV corresponds to the lattice oxygen atoms (O 2− ) in the TiO2; present in the form of oxides, −COO − and C=O in each sample (Fig. 5). No significant change in the binding energies of Ti-2P3/2, Ti-2P1/2 and O-1s is observed in Au-Ag implanted TiO2, which is a clear manifestation of unaffected nano-crystalline structure of TiO2.
After the implantation of Au-Ag in TiO2; very weak Au and Ag peaks present in Au 4f7/2, Au 4f5/2 and Ag-3d5/2, Ag-3d3/2 states appeared at binding energies 83.3, 87.0 eV (Fig. 6) and 367.0, 373.0 eV (Fig. 7), respectively; whose intensity increases with increasing fluence and becomes measurable at A4 and A5 samples. The difference between two states of Au (~3.7 eV) and Ag (~6.0 eV) clearly suggests their presence in zero valent metallic state i.e. Au 0 and Ag 0 27. over, the binding energies of Au-4f5/2 and Ag-3d5/2 are slightly smaller than their metallic state; which arises due to the electron transfer processes taking place from TiO2 to Au and Ag Mnps. The immeasurable Ag and Au atomic %age in A1, A2 and A3 samples indicates Au and Ag content lesser than the 0.5 atomic %age; which is confirmed through decreasing Ti element content. The atomic %age of Ti-2P3/2, Ti-2P1/2, O-1s, Au-4f5/2, Au-4f7/2, Ag-3d5/2 and Ag-3d3/2 elements are summarized in Table 1; showing an increased atomic %age of Ag and Au as well as decreased content of Ti with increased fluence in TiO2.
The UV-visible absorption spectra of TiO2 and Au-Ag embedded TiO2 at different fluence are depicted in Fig. 9(A). TiO2 represents prominent absorption around 310 nm arising due to their intrinsic electronic excitation; with negligible absorption in the visible region of solar spectrum. The range of absorption has been observed to be extended to the visible light region with the implantation of Au and Ag Mnps in TiO2; attributable to the characteristic LSPR absorption peak of Au and Ag lying around 530 nm and 400 nm, respectively stimulated by optical excitation. Also, the LSPR phenomenon of Au and Ag Mnps enhances the absorbance of TiO2 in UV region. The absorbance peak shows a continuous increase with increased fluence up to A3; then a sudden decrement is observed along with the sharp decrease in TiO2 absorbance around 310 nm. It is related to the excessive flattening of TiO2 surface that reduces its absorption cross-sectional area; which is in consonance with FESEM images.
Similar trend has been observed in the absorbance spectra of N719 dye loaded TiO2, A1, A2, A3, A4 and A5 photoanodes and is shown in Fig. 9(B). After dye loading, TiO2 photoanode exhibited strong absorption peaks centered at 400 and 530 nm; which corresponds to the metal to ligand charge transfer interactions within N719 dye molecules. Higher absorbance around 400 nm is due to the merged absorption peaks of TiO2 as well as dye molecules. Broad absorption region extending from UV to NIR light region (300–750 nm) has been observed with the Au-Ag implantation; which is attributed to the synergistic effects of LSPR peaks at 400 and 530 nm associated with Ag and Au Mnps, respectively; that efficiently interacts with dipole moment of dye molecules within a certain minimal spatial range.
The relative change in the absorbance of different Au-Ag implantation fluence based TiO2 photoanodes with respect to unimplanted TiO2 (Fig. 10(A)) for understanding their effective contribution in absorption enhancement in the entire wavelength range; has been estimated from Eq. (1) 25 ;
where, dye concentration is calculated using Beer Lambert’s law at absorbance value around 535 nm, 10 ml volume is taken and effective area is 0.25 cm 2. The variation of dye loading for TiO2, A1, A2, A3, A4 and A5 photoanodes, respectively shown in the inset of Fig. 10(B); suggests that at lower fluence, the dye loading is almost similar but at A4 and A5 fluence it is considerably small; hence confirming lower relative absorbance change in them. Hence, the plasmonic optical effects of Au and Ag Mnps optimally influence the light harvesting ability of TiO2 at A3 fluence based photoanode.
Photovoltaic characteristics of DSSCs
Unimplanted and Au-Ag implanted TiO2 films are used as photoanodes in the fabrication of DSSCs to test their photovoltaic performance under 1 sun illumination conditions (Intensity 100 mW cm −2 ). The photo-current density – voltage (J-V) curves of various DSSCs are shown in Fig. 11; and the obtained short circuit current-density (JSC), open circuit voltage (VOC), fill factor (F.F.) and PCE are tabulated in Table 2. PCE of Au-Ag implanted DSSCs are observed to be higher (A1 (4.69%), A2 (5.49%), A3 (6.56%), A4 (4.18%) and A5 (3.95%)) in comparison to unimplanted DSSC (3.49%); which is due to the enhanced JSC, VOC as well as FF values. Momentous enhancement in JSC of implanted DSSCs (A1 (10.26), A2 (11.90), A3 (14.75), A4 (9.59), and A5 (9.09) mA cm −2 ) relative to unimplanted DSSC (8.78 mA cm −2 ) originates not only from the enhanced light harvesting ability of TiO2, generating enormous amount of photo-excited electrons; but also from the plasmonic electrical effects induced by Au and Ag Mnps embedded in TiO2 photoanodes. The plasmonically excited Mnps also generate hot charge carriers i.e. electrons and fill the trap levels of TiO2, which helps in reducing the charge extraction barrier at C.B. of TiO2 and LUMO level of dye interface that stimulates the transfer of photo-generated electrons to the C.B. of TiO2; hence contributes to the enhanced JSC values in plasmonic DSSCs. Furthermore, Au and Ag Mnps accumulate photo-generated electrons from dye molecules that eventually leads to the upward shifting of Fermi energy level (EF) of TiO2; and has been investigated from Kelvin-Probe work function ( \(\varphi \) ) measurements shown in Fig. 12. It has been observed that \(\varphi \) values decreases in Au-Ag implanted TiO2 relative to unimplanted TiO2. Since, \(\varphi \) is defined as the difference between vacuum energy (EV) and EF level; and EV is constant, EF shifts towards C.B. of TiO2 photoanode; which, further is an indication of increased VOC of implanted TiO2 based DSSCs (A1 (0.66 V), A2 (0.67 V), A3 (0.68 V), A4 (0.65 V) and A4 (0.65 V)) than unimplanted DSSC (0.63 V), as VOC is the difference of EF and redox potential of electrolyte; resulting in higher F.F. values. over, it has been observed that PCE, JSC, VOC and F.F. suddenly decreases at A4 and A5 based DSSC; due to the combined effect of lesser dye loading, generating lesser photo-generated charge carriers as well as higher recombination rate as Au and Ag start acting as charge recombination centers, preventing their transportation to TiO2 C.B. The variations in the photovoltaic parameters of the fabricated DSSCs are presented in Fig. 13. Hence, A3 based DSSC exhibited highest PCE with an augmentation of 87.97% in comparison to the unimplanted DSSCs.
The increased JSC and VOC via upward shifting of EF level due to photo-generated electrons accumulation at Mnps supports the reduction in recombination rate of photo-generated charge carriers in Au-Ag implanted TiO2 photoanodes and is demonstrated by recording PL emission spectra. Since, PL emission arises due to the photons emitted during the recombination of photo-generated electron-hole pairs; offering lesser recombination rate for lower PL intensity. Figure 14 shows broad emissions around 330–550 nm in all the photoanodes; attributable to transitions involving singly ionized oxygen vacancies present in TiO2. A significant decrease is observed in PL intensity of Au-Ag implanted photoanodes; which further decreases with increased fluence up to A3 photoanodes; suggesting effective suppression of recombination rate, as now the photo-generated electrons gets stored at Au and Ag Mnps and hence prolongs the electron lifetime at photoanode, which is later confirmed through EIS measurements. A slight increase at A4 and A5 photoanodes arises due to the agglomeration of Au and Ag in TiO2 letting them act as charge recombination centers.
Figure 15 depicts the Nyquist plots fitted with equivalent circuit model, measured for the unimplanted and Au-Ag implanted DSSCs; to understand their interfacial charge transfer mechanism. It exhibited two semicircles; at higher frequency fitted to resistance offered by reduction reactions at C.E./electrolyte interface (R1); and at intermediate frequency fitted to the charge transfer process at photoanode/electrolyte interface given by charge transfer resistance (R2) and charge transfer capacitance (C) along with a constant phase element (Q). The intercept along X-axis of high frequency semicircle represents equivalent series resistance (RS) having the contribution of FTO, unimplanted and Au-Ag implanted TiO2, Pt CE, and electrolyte of DSSCs; which slightly increases with increasing fluence of Au-Ag Mnps in TiO2 owing to the additional resistance induced by Au and Ag Mnps relative to unimplanted DSSC. R1 is observed to be analogous in all the fabricated DSSCs; because of the usage of same type of Pt CE in them. Substantial increase in R2 values is noticed with the embedment of Au-Ag in TiO2 based DSSCs; with continuous increment for increasing fluence from A1 to A5. It is referred to the hot carrier generation and charge storage ability of Au and Ag Mnps; that lead to decrease in the recombination rate of photo-generated charge carriers; resulting in increasing the electron lifetime at photoanode as well as its transport path length, which leads to efficient charge transportation in DSSCs.
The electron lifetime calculations at TiO2 for all the fabricated DSSCs are investigated through Bode plots (Fig. 16(A)). Fabricated DSSCs exhibited a characteristic frequency maxima, indicating the transient processes occurring at photoanode/electrolyte interface, which shifts towards the lower frequency region with increasing Au-Ag embedment in TiO2; resulting in longer electron lifetime ( \(\tau \) ) calculated from the Eq. (3);
where, fmax is the frequency maxima corresponding to phase shift peak in Bode plots. over, the elongated electron lifetime at photoanode further confirmed the increase in their transport path length. Further, the value of capacitance (C) at C.B. of TiO2 are measured from the expression, \(C=\frac_\) ; which indicates the increased charge storage ability of Au-Ag implanted DSSCs and this capacity enhances with increased fluence of Au-Ag Mnps, as the photo-generated electrons get accumulated on them. Figure 16(B) shows the variation of τ and C with the different fluence of Au-Ag implantation on TiO2 based DSSCs. The EIS parameters such as RS, R2, C, and τ for all the fabricated DSSCs are tabulated in Table 3. Although, Au-Ag embedment in TiO2 facilitates the charge transportation in DSSCs via reducing the recombination, enhancing the electron lifetime as well as charge storage ability; even at highest fluence (A5 based DSSC), still its PCE value is lower which confirmed the sole effect of lower dye loading.
Hence, optimized fluence representing maximum PCE enhancement of highly stable DSSC by 87.97%, due to balanced synergistic interactions involving plasmonic optical and electrical effects of Au and Ag Mnps, is 6 × 10 15 ions cm −2.
Conclusions
In conclusion, we were able to successfully optimize the bimetallic Au-Ag implantation in TiO2 photoanodes for the fabrication of highly efficient third generation DSSCs. Significant enhancement of 87.97% in PCE was achieved for Au-Ag implanted DSSC (Fluence- 6 × 10 15 ions cm −2 ) with JSC and VOC of 14.75 mA cm −2 and 0.68 V, respectively in comparison to unimplanted DSSC (JSC = 8.78 mA cm −2. VOC = 0.63 V and PCE = 3.49%). It is attributed to (i) the enhanced and broadened absorbance of N719 dye sensitizer via simultaneous LSPR property of Au and Ag Mnps along with the hot carrier generation, which improves the light harvesting ability of TiO2 photoanodes; (ii) the reduced recombination reaction rate as Au and Ag Mnps in TiO2 exhibits charge storage ability, leading to increased charge transfer resistances at photoanode/electrolyte interface; (iii) improved interfacial charge carrier transfer processes via increased charge storage capacitance as well as photo-generated electron lifetime.
Materials and Methods
Materials procurement
Materials used for the fabrication of DSSCs such as Fluorine doped tin oxide (FTO), absolute ethanol, zinc powder, hydrochloric acid (HCl), titanium (IV) isopropoxide (TTIP), platinum (Pt) paste and Di-tetrabutylammonium cis-bis(isothiocyanato)bis(2,2′-bipyridyl-4,4′-dicarboxylato)ruthenium(II) (N719) dye with analytical grade quality were purchased from Sigma Aldrich. Titanium dioxide (TiO2) paste and redox electrolyte (iodide-tri iodide \(^/_^\) in 3-methoxypropionitrile (EL-HSE)) were procured from Dyesol, Australia.
Ion Implantation on TiO2 and DSSCs fabrication
Patterned and pre-cleaned FTO substrates, were spin coated with TTIP solution forming a compact layer and were annealed at 450 °C for 30 min. TTIP solution was prepared by adding a solution mixture of 5 ml ethanol and 200 μl HCl into 5 ml ethanol; kept under continuous stirring for 3 hours, along with the drop wise addition of 50 μl TTIP. Further, TiO2 paste was doctor bladed onto the compact layer deposited FTO with effective thickness and area of 3 μm and 0.25 cm 2. respectively; followed by thermal annealing at 450 °C for 30 min. The prepared TiO2 films were implanted firstly with Ag ion beam (Energy-120 KeV) at room temperature under high vacuum conditions using low energy negative ion implanter (Inter University Accelerator Centre (IUAC), New Delhi, India) at different fluence of 10 15 –10 16 ions cm −2. Secondly, Au ion beam was implanted with energy 80 KeV onto Ag implanted TiO2 at respective similar fluence. The Au-Ag implanted TiO2 films are named as A1, A2, A3, A4 and A5 for 1 × 10 15. 3 × 10 15. 6 × 10 15. 9 × 10 15 and 1.2×10 16 ions cm −2. respectively. Au and Ag penetrated up to 22 and 17 nm depth in TiO2 and are calculated using the stopping and range of ions in matter (SRIM) software. The unimplanted and Au-Ag implanted TiO2 were immersed into N719 dye (0.3 mM concentration) for 24 h. Pt was deposited on pre-cleaned FTO substrates and were annealed at 450 °C for 30 min. for the preparation of counter electrode (CE). DSSCs were assembled by sandwiching different photoanodes and Pt CE using redox electrolyte in them as an intermediate to complete the circuit.
Characterizations
Crystal structure of unimplanted and Au-Ag implanted TiO2 films were determined with X-ray diffraction (XRD) using D8 FOCUS, Bruker Ettlingen with Cu Kα radiation (λ = 1.5418 Å, Current = 30 mA and Voltage = 40 kV) from 5–80° Bragg’s angle. X-ray photoelectron spectroscopy (XPS) was done for elemental analysis via MAC2 electron analyzer system interconnected with MBE machine (EVA-32 Riber, France) at an excitation energy of 1253.3 eV using Mg Kα X-ray beam; within binding energy (B.E.) range 10–1500 eV. System used was initially calibrated using Au 4f7/2 line with 84.0 eV B.E. Surface morphology of the prepared films were investigated using field emission scanning electron microscope (FESEM-Carl Zeiss, Supra 55). UV-Vis measurements were done using SHIMADZU, UV-VIS NIR 3600 spectrometer within 250–800 nm wavelength range. To perform the desorption experiments, the N719 dye was desorbed from unimplanted and Au-Ag implanted TiO2 using 0.1 M aqueous solution of potassium hydroxide (KOH) and further, the absorbance spectra of deloaded dye solutions were recorded. Photovoltaic parameters of fabricated DSSCs were studied using Keithley source meter (Model 2400) under 1 sun illumination at 1.5 G AM of intensity 100 mW cm −2 with OAI, TriSOL solar simulator; which was initially calibrated with standard silicon cell prior to the measurements. The work function measurements of unimplanted and Au-Ag implanted TiO2 were recorded using Kelvin Probe technique (SKP, Kelvin Probe 4.5). The photoluminescence (PL) spectra was obtained at an excitation wavelength of 310 nm using PerkinElmer, LS 55 Fluorescence Spectrometer. Electrochemical impedance spectroscopy (EIS) was performed using frequency response analyzer (FRA) connected with Autolab potentiostat/galvanostat (PGSTAT12) within the frequency range of 0.1 Hz to 1.0 MHz 19,20.
Acknowledgements
One of the authors, Navdeep Kaur, is grateful to UGC, New Delhi, India for providing financial support to do the present research work. The authors acknowledge Inter University Accelerator Centre, New Delhi for providing beam times through Project No. UFR-60331. The authors are sincerely thankful to BARC, Mumbai for providing XPS and Kelvin Probe Measurements.
Author information
Authors and Affiliations
- Department of Physics, Guru Nanak Dev University, Amritsar, 143 005, India Navdeep Kaur, Viplove Bhullar, Davinder Paul Singh Aman Mahajan
Perovskite Solar
Perovskites are a class of materials that share a similar structure, which display a myriad of exciting properties like superconductivity, magnetoresistance and more. These easily synthesized materials are considered the future of solar cells, as their distinctive structure makes them perfect for enabling low-cost, efficient photovoltaics. They are also predicted to play a role in next-gen electric vehicle batteries, sensors, lasers and much more.
How does the PV market look today?
In general, Photovoltaic (PV) technologies can be viewed as divided into two main categories: wafer-based PV (also called 1st generation PVs) and thin-film cell PVs. Traditional crystalline silicon (c-Si) cells (both single crystalline silicon and multi-crystalline silicon) and gallium arsenide (GaAs) cells belong to the wafer-based PVs, with c-Si cells dominating the current PV market (about 90% market share) and GaAs exhibiting the highest efficiency.
Thin-film cells normally absorb light more efficiently than silicon, allowing the use of extremely thin films. Cadmium telluride (CdTe) technology has been successfully commercialized, with more than 20% cell efficiency and 17.5% module efficiency record and such cells currently hold about 5% of the total market. Other commercial thin-film technologies include hydrogenated amorphous silicon (a-Si:H) and copper indium gallium (di)selenide (CIGS) cells, taking approximately 2% market share each today. Copper zinc tin sulphide technology has been under RD for years and will probably require some time until actual commercialization.
What is a perovskite solar cell?
An emerging thin-film PV class is being formed, also called 3rd generation PVs, which refers to PVs using technologies that have the potential to overcome current efficiency and performance limits or are based on novel materials. This 3rd generation of PVs includes DSSC, organic photovoltaic (OPV), quantum dot (QD) PV and perovskite PV.
A perovskite solar cell is a type of solar cell which includes a perovskite structured compound, most commonly a hybrid organic-inorganic lead or tin halide-based material, as the light-harvesting active layer. Perovskite materials such as methylammonium lead halides are cheap to produce and relatively simple to manufacture. Perovskites possess intrinsic properties like broad absorption spectrum, fast charge separation, long transport distance of electrons and holes, long carrier separation lifetime, and more, that make them very promising materials for solid-state solar cells.
Perovskite solar cells are, without a doubt, the rising star in the field of photovoltaics. They are causing excitement within the solar power industry with their ability to absorb light across almost all visible wavelengths, exceptional power conversion efficiencies already exceeding 20% in the lab, and relative ease of fabrication. Perovskite solar cells still face several challenge, but much work is put into facing them and some companies, are already talking about commercializing them in the near future.
What are the advantages of Perovskite solar cells?
Put simply, perovskite solar cells aim to increase the efficiency and lower the cost of solar energy. Perovskite PVs indeed hold promise for high efficiencies, as well as low potential material reduced processing costs. A big advantage perovskite PVs have over conventional solar technology is that they can react to various different wavelengths of light, which lets them convert more of the sunlight that reaches them into electricity.
over, they offer flexibility, semi-transparency, tailored form factors, light-weight and more. Naturally, electronics designers and researchers are certain that such characteristics will open up many more applications for solar cells.
What is holding perovskite PVs back?
Despite its great potential, perovskite solar cell technology is still in the early stages of commercialization compared with other mature solar technologies as there are a number of concerns remaining.
One problem is their overall cost (for several reasons, mainly since currently the most common electrode material in perovskite solar cells is gold), and another is that cheaper perovskite solar cells have a short lifespan. Perovskite PVs also deteriorate rapidly in the presence of moisture and the decay products attack metal electrodes. Heavy encapsulation to protect perovskite can add to the cell cost and weight. Scaling up is another issue. reported high efficiency ratings have been achieved using small cells, which is great for lab testing, but too small to be used in an actual solar panel.
A major issue is toxicity. a substance called PbI is one of the breakdown products of perovskite. This is known to be toxic and there are concerns that it may be carcinogenic (although this is still an unproven point). Also, many perovskite cells use lead, a massive pollutant. Researchers are constantly seeking substitutions, and have already made working cells using tin instead. (with efficiency at only 6%, but improvements will surely follow).
What’s next?
While major challenges indeed exist, perovskite solar cells are still touted as the PV technology of the future, and much development work and research are put into making this a reality. Scientists and companies are working towards increasing efficiency and stability, prolonging lifetime and replacing toxic materials with safer ones. Researchers are also looking at the benefits of combining perovskites with other technologies, like silicon for example, to create what is referred to as “tandem cells”.
Commercial activity in the field of perovskite PV
In September 2015, Australia-based organic PV and perovskite solar cell (PSC) developer Dyesol declared a major breakthrough in perovskite stability for solar applications. Dyesol claims to have made a significant breakthrough on small perovskite solar cells, with “meaningful numbers” of 10% efficient strip cells exhibiting less than 10% relative degradation when exposed to continuous light soaking for over 1000 hours. Dyesol was also awarded a 0.5 million grant from the Australian Renewable Energy Agency (ARENA) to commercialize an innovative, very high efficiency perovskite solar cell.
Also in 2015, Saule Technologies signed an investment deal with Hideo Sawada, a Japanese investment company. Saule aims to combine perovskite solar cells with other currently available products, and this investment agreement came only a year after the company was launched.
In October 2020, Saule launched sunbreaker lamellas equipped with perovskite solar cells. The product is planned to soon be marketed across across Europe and potentially go global after that.
In August 2020, reports out of China suggested that a perovskite photovoltaic cell production line has gone into production in Quzhou, east China’s Zhejiang Province. The 40-hectare factory was reportedly funded by Microquanta Semiconductor and expected to produce more than 200,000 square meters of photovoltaic glass before the end of 2020.
EneCoat Technologies and Toyota to jointly develop automotive perovskite solar cells
EneCoat Technologies and Toyota Motor Corporation have announced that they will work together to develop and commercialize automotive perovskite solar cells (PSCs) with the common goal of contributing to the realization of carbon neutrality. The high efficiency, thin form factor and light weight make PSCs suitable for the automotive industry.
EneCoat is a start-up company established in 2018 based on research results from Atsushi Wakamiya’s laboratory at the Institute for Chemical Research, Kyoto University. It has developed material and deposition technologies for high-efficiency perovskite solar cells, and has successfully developed film-type perovskite solar cells with high output (module conversion efficiency of 19.4% as of April 2023). It is also participating in the Green Innovation Fund Project, one of the government’s industrial policies aiming for carbon neutrality by 2050.
Researchers suggest substitute for gold to make perovskite solar cells more affordable
Researchers from Northern Illinois University, National Renewable Energy Laboratory (NREL), Northwestern University and Argonne National Laboratory have reported a bilayer back electrode configuration consisting of an Ni-doped natural graphite layer with a fusible Bi-In alloy. This back electrode can be deposited in a vacuum-free approach and enables perovskite solar cells (PSCs) with a power conversion efficiency of 21.0%. These inexpensive materials and facile ambient fabrication techniques can help provide an appealing solution to low-cost PSC industrialization.
A thin layer of gold or silver can help improve the efficiency of perovskite solar cells, but the researchers have found a less expensive material that will enable commercialization of the technology without exorbitant cost. “A layer of gold in a solar panel or even a layer of silver is probably too expensive,” said Kai Zhu, a senior scientist in the Chemistry and Nanoscience Center at the U.S. Department of Energy’s National Renewable Energy Laboratory (NREL). “It would make the solar panel not affordable for most people.”
Researchers announce 24.35% efficiency for inverted perovskite solar cells on an active area of 1 cm2
Researchers at the National University of Singapore (NUS) and Solar Energy Research Institute of Singapore (SERIS) have announced achieving 24.35% efficiency for self-designed inverted perovskite solar cells on an active area of 1 cm 2. saying it is an improvement over the previous record high of 23.7% on the same area.
To get to this 24.35% efficiency level, the team says it incorporated a novel interface material into perovskite cells that contributed a ‘range of advantageous attributes’. These include excellent optical, electrical and chemical properties that enhanced both their efficiency and longevity.
Researchers examine perovskite-sensitized upconversion under real-world conditions
Researchers at Florida State University, the FAMU-FSU College of Engineering, the University of Colorado Boulder and Argonne National Laboratory have studied the effects of two stressors, heat and light, on the triplet generation process at the perovskite/rubrene interface. Following exposure to both stressors, local discrepancies across the upconversion device were discovered. This work emphasizes the challenges and continued potential for the integration of perovskite-sensitized upconversion (UC) into commercial photovoltaic devices.
The first region showed changes to the morphology, and no detectable upconverted emission was observed. Through the combination of optical microscopy and spectroscopy, crystallization of the organic semiconductor layer, degradation of dibenzotetraphenylperiflanthene, and concurrent degradation of the perovskite sensitizer were found. These effects culminate in a reduction in both triplet generation and triplet–triplet annihilation. In the second region, no changes to the morphology were present and visible UC emission was observed following exposure to both stressors. To probe the triplet sensitization process at elevated temperatures, transient absorption spectroscopy was performed. The presence of the excited spin-triplet state of rubrene at 60 °C highlighted successful triplet generation even at elevated temperatures.
Researchers find that temperature coefficient may not a proper metric for perovskite/silicon tandem solar cells
Researchers from Saudi Arabia’s King Abdullah University of Science and Technology (KAUST) have conducted a series of tests to see if the temperature coefficient of the short circuit current in perovskite-silicon tandem solar cells could be a proper standard of measurement to analyze their behavior and performance. The team has come to the conclusion that it may not be considered a proper metric to assess these devices’ performance and behavior.
The scientists explained that the idea of their recent paper was to show that the temperature coefficient of the short circuit current, measured under standard illumination conditions, might not well describe the actual operation of tandem solar cells. Depending on the local spectrum and the temperature range, the current at maximum power (Impp) can increase, decrease or have a mixed behavior. This intriguing effect can help the community to better understand their outdoor data as outdoor tandem operation should become more and more common in the next years.
Perovskite PV modules. affordable and efficient. are available now!
This is a sponsored message by Solaires
Solaires is recycling indoor light to power IoT and electronic devices
Solaires Entreprises Inc. is a Canadian solar energy startup based in Victoria, BC., who has developed perovskite photovoltaic (PV) modules, designed for integration into IoT devices, small consumer electronics, and Smart gadgets. Powered by indoor light, the cells are extremely efficient, modular, and are configurable to suit the application.
For years, the consumer electronics industry has dreamt of new technologies to power or recharge devices with indoor light. Perovskite modules are the most suitable solution and Solaires is proud to announce their perovskite PV modules are now available to the market for evaluation and integration into your devices! The company offers custom module sizes tailored to perfectly suit your needs. Those familiar in this space will appreciate that Solaires PV modules provide superior light absorption to traditional materials.
Why perovskite technology?
Theoretically, the best absorber materials for indoor applications should have an energy Band gap between 1.8 to 2.0 eV. Silicon, with a bandgap between 1.1 to 1.6 eV shows poor performance for indoor light. Perovskite, though, has a tunable bandgap. Solaires’ team can adjust the bandgap to be between 1.2 to 2.6 eV, making perovskite PV modules suitable for generating high power from indoor light. This is achieved by a simple and cost effective solution processed to engineer the perovskite composition and the resulting Band gap. As a result, Solaires can make perovskite PV modules suitable for generating high power from indoor light.
Fig. 1 illustrates the efficiency of perovskite, note the red area signalling the bandgap for electric light.
National University of Singapore and REC Solar launch US57.4 million solar research initiative
The National University of Singapore (NUS) and REC Solar, the solar power arm of the Singaporean Renewable Energy Corporation, have launched a S77 million (US57.4 million) solar cell research initiative.
The project, called the REC@NUS Corporate RD Laboratory for Next Generation Photovoltaics, will be led by two co-directors, professor Amrin Aberle, CEO of the NUS’ Solar Energy Research Institute of Singapore, and Shankar G Sridhara, chief technology officer of REC Solar. The university announced that the initiative will develop and commercialize “disruptive” PV technologies, based on perovskite-silicon tandem solar cells in particular.
Researchers design perovskite solar cell with 20.15% efficiency using indium sulfide ETL
An international team has designed a perovskite solar cell with an electron transport layer (ETL) made of indium sulfide (In2S3) that could reportedly reduce the defect density in the device.
The scientists used a numerical module and the SCAPS-1D solar cell capacitance software, which is a simulation tool for thin-film solar cells that was developed by the University of Ghent in Belgium, to simulate s solar cell based on methylammonium lead iodide (MAPbI3).
TCI’s molecular dopants boost organic electronics
TCI has launched a range of molecular dopants that can significantly increase the charge carrier density and modify the energy levels in organic electronics devices. Molecular dopants offer a versatile platform to tune the optoelectrical and electrical properties of organic semiconductors to application-specific demands, allowing advantages like increasing the electrical conductivity and mobility by orders of magnitude and improving contact properties in various electronic and optoelectronic devices.
TCI’s p-type and n-type dopants can be applied to various organic electronics devices, such as: carrier transport layers of organic light-emitting diodes (OLEDs), organic photovoltaics (OPVs), perovskite solar cells (PSCs), and perovskite quantum dot LEDs, as well as active layers of organic field-effect transistors (OFETs), OPVs, and thermoelectric devices in the field of organic electronics research.
LONGi announces conversion efficiency of 33.5% for silicon-perovskite tandem solar cells based on commercial CZ silicon wafers
LONGi announced new conversion efficiency of 33.5% for silicon-perovskite tandem solar cells based on commercial CZ silicon wafers, as was revealed at the Intersolar Europe 2023 exhibition. Earlier this month, LONGi announced its “STAR Innovative Ecological Cooperation Platform” and its newly achieved efficiency of 31.8% for perovskite/crystalline silicon tandem solar cells based on commercial CZ silicon wafers.
“Improving cell conversion efficiency and reducing the cost of electricity remain the perpetual theme driving the development of the photovoltaic industry.” Said Li Zhenguo, President of LONGi.
Solar Photovoltaic Technology Basics
Solar cells, also called photovoltaic cells, convert sunlight directly into electricity.
Photovoltaics (often shortened as PV) gets its name from the process of converting light (photons) to electricity (voltage), which is called the photovoltaic effect. This phenomenon was first exploited in 1954 by scientists at Bell Laboratories who created a working solar cell made from silicon that generated an electric current when exposed to sunlight. Solar cells were soon being used to power space satellites and smaller items such as calculators and watches. Today, electricity from solar cells has become cost competitive in many regions and photovoltaic systems are being deployed at large scales to help power the electric grid.
Silicon Solar Cells
The vast majority of today’s solar cells are made from silicon and offer both reasonable and good efficiency (the rate at which the solar cell converts sunlight into electricity). These cells are usually assembled into larger modules that can be installed on the roofs of residential or commercial buildings or deployed on ground-mounted racks to create huge, utility-scale systems.
Thin-Film Solar Cells
Another commonly used photovoltaic technology is known as thin-film solar cells because they are made from very thin layers of semiconductor material, such as cadmium telluride or copper indium gallium diselenide. The thickness of these cell layers is only a few micrometers—that is, several millionths of a meter.
Thin-film solar cells can be flexible and lightweight, making them ideal for portable applications—such as in a soldier’s backpack—or for use in other products like Windows that generate electricity from the sun. Some types of thin-film solar cells also benefit from manufacturing techniques that require less energy and are easier to scale-up than the manufacturing techniques required by silicon solar cells.
III-V Solar Cells
A third type of photovoltaic technology is named after the elements that compose them. III-V solar cells are mainly constructed from elements in Group III—e.g., gallium and indium—and Group V—e.g., arsenic and antimony—of the periodic table. These solar cells are generally much more expensive to manufacture than other technologies. But they convert sunlight into electricity at much higher efficiencies. Because of this, these solar cells are often used on satellites, unmanned aerial vehicles, and other applications that require a high ratio of power-to-weight.
Next-Generation Solar Cells
Solar cell researchers at NREL and elsewhere are also pursuing many new photovoltaic technologies—such as solar cells made from organic materials, quantum dots, and hybrid organic-inorganic materials (also known as perovskites). These next-generation technologies may offer lower costs, greater ease of manufacture, or other benefits. Further research will see if these promises can be realized.
Reliability and Grid Integration Research
Photovoltaic research is more than just making a high-efficiency, low-cost solar cell. Homeowners and businesses must be confident that the solar panels they install will not degrade in performance and will continue to reliably generate electricity for many years. Utilities and government regulators want to know how to add solar PV systems to the electric grid without destabilizing the careful balancing act between electricity supply and demand.
Materials scientists, economic analysts, electrical engineers, and many others at NREL are working to address these concerns and ensure solar photovoltaics are a clean and reliable source of energy.
Additional Resources
For more information about solar photovoltaic energy, visit the following resources:
Solar Photovoltaic Technology BasicsU.S. Department of Energy’s Office of Energy Efficiency and Renewable Energy